Search
- Page Path
- HOME > Search
Short Communication
- Epidemiological characteristics of carbapenemase-producing Enterobacteriaceae outbreaks in the Republic of Korea between 2017 and 2022
- Hyoseon Jeong, Junghee Hyun, Yeon-Kyeng Lee
- Osong Public Health Res Perspect. 2023;14(4):312-320. Published online August 21, 2023
- DOI: https://doi.org/10.24171/j.phrp.2023.0069
- 1,097 View
- 144 Download
- 2 Crossref
-
 Graphical Abstract
Graphical Abstract
 Abstract
Abstract
 PDF
PDF 
- Objectives
We aimed to describe the epidemiological characteristics of carbapenemase-producing Enterobacteriaceae (CPE) outbreaks in healthcare settings in the Republic of Korea between 2017 and 2022.
Methods
Under the national notifiable disease surveillance system, we obtained annual descriptive statistics regarding the isolated species, carbapenemase genotype, healthcare facility type, outbreak location and duration, and number of patients affected and recommended interventions. We used epidemiological investigation reports on CPE outbreaks reported to Korea Disease Control and Prevention Agency from June 2017 to September 2022.
Results
Among the 168 reports analyzed, Klebsiella pneumoniae (85.1%) was the most frequently reported species, while K. pneumoniae carbapenemase (KPC, 82.7%) was the most common carbapenemase genotype. Both categories increased from 2017 to 2022 (p<0.01). General hospitals had the highest proportion (54.8%), while tertiary general hospitals demonstrated a decreasing trend (p<0.01). The largest proportion of outbreaks occurred exclusively in intensive care units (ICUs, 44.0%), and the frequency of concurrent outbreaks in ICUs and general wards increased over time (p<0.01). The median outbreak duration rose from 43.5 days before the coronavirus disease 2019 (COVID-19) pandemic (2017–2019) to 79.5 days during the pandemic (2020–2022) (p=0.01), and the median number of patients associated with each outbreak increased from 5.0 to 6.0 (p=0.03). Frequently recommended interventions included employee education (38.1%), and 3 or more measures were proposed for 45.2% of outbreaks.
Conclusion
In the Republic of Korea, CPE outbreaks have been consistently dominated by K. pneumoniae and KPC. The size of these outbreaks increased during the COVID-19 pandemic. Our findings highlight the need for continuing efforts to control CPE outbreaks using a multimodal approach, while considering their epidemiology. -
Citations
Citations to this article as recorded by- Comparison of clinical outcomes of patients with serial negative surveillance cultures according to a subsequent polymerase chain reaction test for carbapenemase-producing Enterobacterales
H. Seo, S. Kim, Y.W. Lee, H.S. Oh, H-S. Kim, Y.K. Kim
Journal of Hospital Infection.2024; 146: 93. CrossRef - Identifying Contact Time Required for Secondary Transmission of Clostridioides difficile Infections by Using Real-Time Locating System
Min Hyung Kim, Jaewoong Kim, Heejin Ra, Sooyeon Jeong, Yoon Soo Park, Dongju Won, Hyukmin Lee, Heejung Kim
Emerging Infectious Diseases.2024;[Epub] CrossRef
- Comparison of clinical outcomes of patients with serial negative surveillance cultures according to a subsequent polymerase chain reaction test for carbapenemase-producing Enterobacterales
Brief Reports
- Adverse events of the Pfizer-BioNTech COVID-19 vaccine in Korean children and adolescents aged 5 to 17 years
- Seontae Kim, Yeseul Heo, Soon-Young Seo, Do Sang Lim, Enhi Cho, Yeon-Kyeng Lee
- Osong Public Health Res Perspect. 2022;13(5):382-390. Published online October 14, 2022
- DOI: https://doi.org/10.24171/j.phrp.2022.0233
- 2,394 View
- 111 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - Objectives
This study aimed to identify potential safety signals and adverse events following the primary Pfizer-BioNTech coronavirus disease 2019 (COVID-19) vaccination series among children and adolescents aged 5 to 17 years in the Republic of Korea. Methods: Adverse events reported through the COVID-19 vaccination management system (CVMS, a web-based passive vaccine safety surveillance system) and adverse events and health conditions collected from a text message-based survey were analyzed. Results: A total of 14,786 adverse events among 5 to 17-year-old children and adolescents were reported in the CVMS; 14,334 (96.9%) were non-serious and 452 (3.1%) were serious, including 125 suspected cases of acute cardiovascular injury and 101 suspected cases of anaphylaxis. The overall reporting rate was lower in 5 to 11-year-old children (64.5 per 100,000 doses) than in 12 to 17-year-old adolescents (300.5 per 100,000 doses). The text message survey identified that local and systemic adverse events after either dose were reported less frequently in 5 to 11-year-old children than in 12 to 17-year-old adolescents (p<0.001). The most commonly reported adverse events were pain at the injection site, myalgia, headache, and fatigue/tiredness. Conclusion: The overall results are consistent with the results of controlled trials; serious adverse events were extremely rare among 5 to 17-year-old children and adolescents following Pfizer-BioNTech COVID-19 vaccination. Adverse events were less frequent in children aged 5 to 11 years than in adolescents aged 12 to 17 years. -
Citations
Citations to this article as recorded by- Safety monitoring of COVID-19 vaccines: February 26, 2021, To June 4, 2022, Republic of Korea
Yeon-Kyeng Lee, Yunhyung Kwon, Yesul Heo, Eun Kyoung Kim, Seung Yun Kim, Hoon Cho, Seontae Kim, Mijeong Ko, Dosang Lim, Soon-Young Seo, Enhi Cho
Clinical and Experimental Pediatrics.2023; 66(10): 415. CrossRef - Effectiveness of the BNT162b2 vaccine in preventing morbidity and mortality associated with COVID-19 in children aged 5 to 11 years: A systematic review and meta-analysis
Sumayyah Ebrahim, Ntombifuthi Blose, Natasha Gloeck, Ameer Hohlfeld, Yusentha Balakrishna, Rudzani Muloiwa, Andy Gray, Andy Parrish, Karen Cohen, Ruth Lancaster, Tamara Kredo, Julia Robinson
PLOS Global Public Health.2023; 3(12): e0002676. CrossRef
- Safety monitoring of COVID-19 vaccines: February 26, 2021, To June 4, 2022, Republic of Korea
- Safety monitoring of COVID-19 vaccination among adolescents aged 12 to 17 years old in the Republic of Korea
- Seontae Kim, Insob Hwang, Mijeong Ko, Yunhyung Kwon, Yeon-Kyeng Lee
- Osong Public Health Res Perspect. 2022;13(3):230-237. Published online June 10, 2022
- DOI: https://doi.org/10.24171/j.phrp.2022.0122
- 4,145 View
- 139 Download
- 6 Web of Science
- 7 Crossref
-
 Graphical Abstract
Graphical Abstract
 Abstract
Abstract
 PDF
PDF 
- Objectives
This study aimed to disseminate information on coronavirus disease 2019 (COVID-19) vaccine safety among adolescents aged 12 to 17 years in the Republic of Korea. Methods: Two databases were used to assess COVID-19 vaccine safety in adolescents aged 12 to 17 years who completed the primary Pfizer-BioNTech vaccination series. Adverse events reported to the web-based COVID-19 vaccination management system (CVMS) and collected in the text message-based system were analyzed. Results: From March 5, 2021 to February 13, 2022, 12,216 adverse events among 12- to 17-yearolds were reported to the CVMS, of which 97.1% were non-serious adverse events and 2.9% were serious adverse events, including 85 suspected cases of anaphylaxis, 74 suspected cases of myocarditis and/or pericarditis, and 2 deaths. From December 13, 2021 to January 26, 2022, 10,389 adolescents responded to a text message survey, and local/systemic adverse events were more common after dose 2 than after dose 1. The most commonly reported events following either vaccine dose were pain at the injection site, headache, fatigue/tiredness, and myalgia. Conclusion: The overall results are consistent with previous findings; the great majority of adverse events were non-serious, and serious adverse events were rare among adolescents aged 12 to 17 years following Pfizer-BioNTech COVID-19 vaccination. -
Citations
Citations to this article as recorded by- Suspected Myocarditis after mRNA COVID-19 Vaccination among South Korean Adolescents
Mi Jin Kim, Jin Hee Kim, Hyun Ok Jun, Kyung Min Kim, Min Sub Jeung, Jun Sung Park
Journal of Pediatric Infectious Diseases.2024; 19(02): 075. CrossRef - Characterization of Brighton Collaboration criteria for myocarditis and pericarditis following COVID-19 vaccine in Korean adolescents
Jue Seong Lee, HyoSug Choi, Seung Hwan Shin, Myung-Jae Hwang, Sara Na, Jong Hee Kim, Sangshin Park, Yoonsun Yoon, Hyun Mi Kang, Bin Ahn, Kyoungsan Seo, Young June Choe
Vaccine.2024;[Epub] CrossRef - Immunogenicity, effectiveness, and safety of COVID-19 vaccines among children and adolescents aged 2–18 years: an updated systematic review and meta-analysis
Peng Gao, Liang-Yu Kang, Jue Liu, Min Liu
World Journal of Pediatrics.2023; 19(11): 1041. CrossRef - Incidence of myopericarditis after mRNA COVID-19 vaccination: A meta-analysis with focus on adolescents aged 12–17 years
Bao-Qiang Guo, Hong-Bin Li, Li-Qiang Yang
Vaccine.2023; 41(28): 4067. CrossRef - Safety monitoring of COVID-19 vaccines: February 26, 2021, To June 4, 2022, Republic of Korea
Yeon-Kyeng Lee, Yunhyung Kwon, Yesul Heo, Eun Kyoung Kim, Seung Yun Kim, Hoon Cho, Seontae Kim, Mijeong Ko, Dosang Lim, Soon-Young Seo, Enhi Cho
Clinical and Experimental Pediatrics.2023; 66(10): 415. CrossRef - Risk of Coronavirus Disease 2019 Messenger RNA Vaccination-Associated Myocarditis and Pericarditis – A Systematic Review of Population-Based Data
Yen-Ching Lin, Chia-Hsuin Chang, Wei-Ju Su, Chin-Hui Yang, Jann-Tay Wang
Risk Management and Healthcare Policy.2023; Volume 16: 2085. CrossRef - COVID-19 Vaccination in Korea: Past, Present, and the Way Forward
Eliel Nham, Joon Young Song, Ji Yun Noh, Hee Jin Cheong, Woo Joo Kim
Journal of Korean Medical Science.2022;[Epub] CrossRef
- Suspected Myocarditis after mRNA COVID-19 Vaccination among South Korean Adolescents
- COVID-19 vaccine safety monitoring in Republic of Korea from February 26, 2021 to October 31, 2021
- Insob Hwang, Kyeongeun Park, Tae Eun Kim, Yunhyung Kwon, Yeon-Kyeng Lee
- Osong Public Health Res Perspect. 2021;12(6):396-402. Published online December 21, 2021
- DOI: https://doi.org/10.24171/j.phrp.2021.0310
- 7,225 View
- 191 Download
- 13 Web of Science
- 11 Crossref
-
 Graphical Abstract
Graphical Abstract
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material 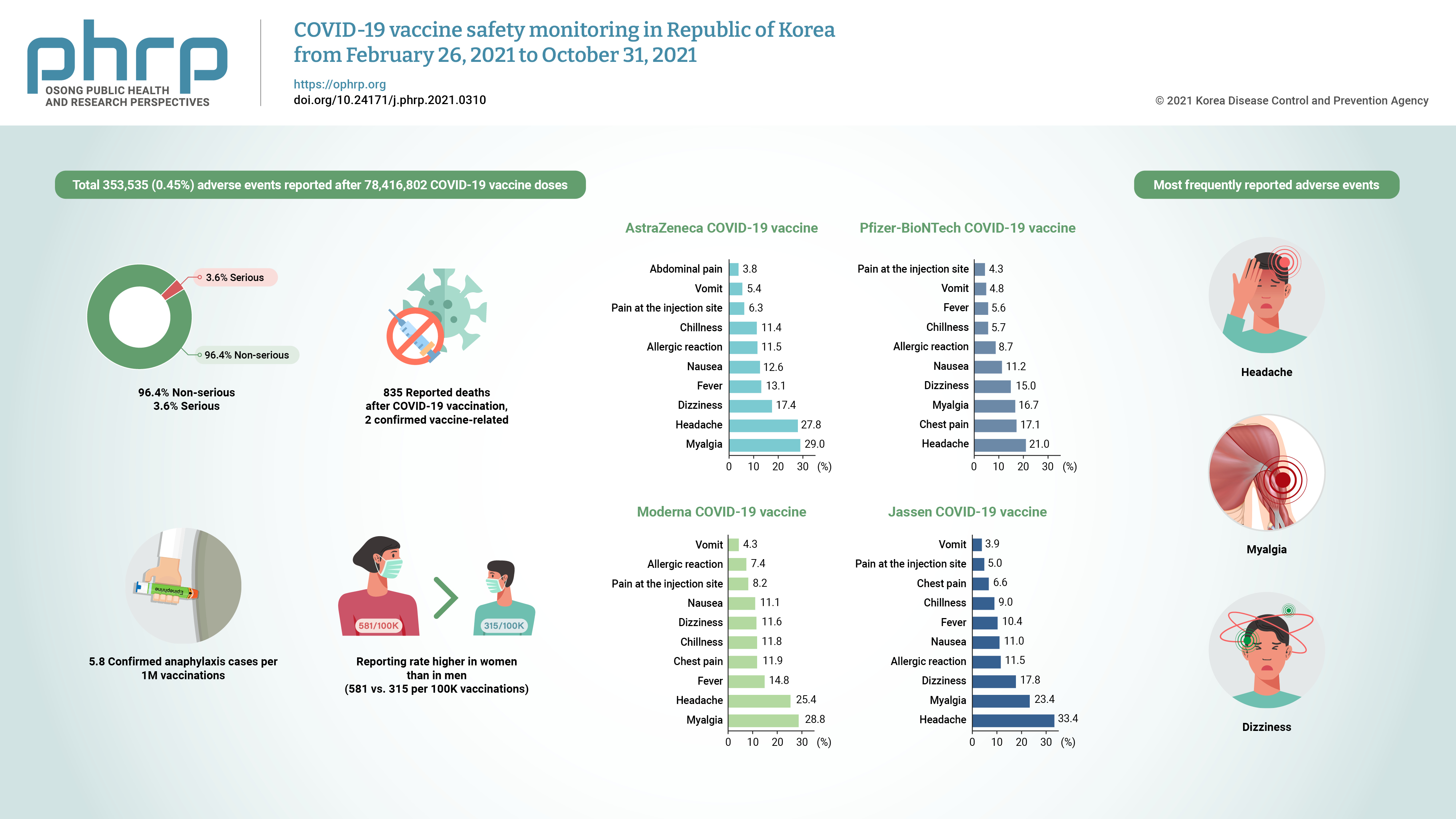
- Objectives
This study aimed to present data on reported adverse events following coronavirus disease 2019 (COVID-19) vaccination in Republic of Korea from February 26 to October 31, 2021, and to determine whether any significant patterns emerged from an analysis of the characteristics of suspected adverse event cases for each type of vaccine.
Methods
Adverse events following COVID-19 vaccination reported by medical doctors and forensic pathologists were analyzed. Cases of suspected anaphylaxis were classified using the Brighton Collaboration definition.
Results
By October 31, 2021, a total of 353,535 (0.45%) adverse events were reported after 78,416,802 COVID-19 vaccine doses. Of the adverse events, 96.4% were non-serious and 3.6% were serious. The most frequently reported adverse events were headache, myalgia, and dizziness. Of the 835 reported deaths after COVID-19 vaccination, 2 vaccine-related deaths were confirmed. Suspected anaphylaxis was confirmed in 454 cases using the Brighton Collaboration definition.
Conclusion
The commonly reported symptoms were similar to those described in clinical trials. Most reported adverse events were non-serious, and the reporting rate of adverse events following COVID-19 vaccination was higher in women than in men (581 vs. 315 per 100,000 vaccinations). Confirmed anaphylaxis was reported in 5.8 cases per 1,000,000 vaccinations. -
Citations
Citations to this article as recorded by- A Nationwide Survey of mRNA COVID-19 Vaccinee’s Experiences on Adverse Events and Its Associated Factors
Dongwon Yoon, Ha-Lim Jeon, Yunha Noh, Young June Choe, Seung-Ah Choe, Jaehun Jung, Ju-Young Shin
Journal of Korean Medical Science.2023;[Epub] CrossRef - Temporal association between the age-specific incidence of Guillain-Barré syndrome and SARS-CoV-2 vaccination in Republic of Korea: a nationwide time-series correlation study
Hyunju Lee, Donghyok Kwon, Seoncheol Park, Seung Ri Park, Darda Chung, Jongmok Ha
Osong Public Health and Research Perspectives.2023; 14(3): 224. CrossRef - Safety monitoring of COVID-19 vaccines: February 26, 2021, To June 4, 2022, Republic of Korea
Yeon-Kyeng Lee, Yunhyung Kwon, Yesul Heo, Eun Kyoung Kim, Seung Yun Kim, Hoon Cho, Seontae Kim, Mijeong Ko, Dosang Lim, Soon-Young Seo, Enhi Cho
Clinical and Experimental Pediatrics.2023; 66(10): 415. CrossRef - Allergic Reactions to COVID-19 Vaccines: Risk Factors, Frequency, Mechanisms and Management
Nicoletta Luxi, Alexia Giovanazzi, Alessandra Arcolaci, Patrizia Bonadonna, Maria Angiola Crivellaro, Paola Maria Cutroneo, Carmen Ferrajolo, Fabiana Furci, Lucia Guidolin, Ugo Moretti, Elisa Olivieri, Giuliana Petrelli, Giovanna Zanoni, Gianenrico Senna,
BioDrugs.2022; 36(4): 443. CrossRef - Safety monitoring of COVID-19 vaccination among adolescents aged 12 to 17 years old in the Republic of Korea
Seontae Kim, Insob Hwang, Mijeong Ko, Yunhyung Kwon, Yeon-Kyeng Lee
Osong Public Health and Research Perspectives.2022; 13(3): 230. CrossRef - Incidence and Characteristics of Adverse Events after COVID-19 Vaccination in a Population-Based Programme
Laura Bonzano, Olivera Djuric, Pamela Mancuso, Lidia Fares, Raffaele Brancaccio, Marta Ottone, Eufemia Bisaccia, Massimo Vicentini, Alessia Cocconcelli, Alfonso Motolese, Rostyslav Boyko, Paolo Giorgi Rossi, Alberico Motolese
Vaccines.2022; 10(7): 1111. CrossRef - Global Predictors of COVID-19 Vaccine Hesitancy: A Systematic Review
Carla Pires
Vaccines.2022; 10(8): 1349. CrossRef - Anaphylaxis and Related Events Following COVID‐19 Vaccination: A Systematic Review
Pradipta Paul, Emmad Janjua, Mai AlSubaie, Vinutha Ramadorai, Beshr Mushannen, Ahamed Lazim Vattoth, Wafa Khan, Khalifa Bshesh, Areej Nauman, Ibrahim Mohammed, Imane Bouhali, Mohammed Khalid, Dalia Zakaria
The Journal of Clinical Pharmacology.2022; 62(11): 1335. CrossRef - Adverse events of the Pfizer-BioNTech COVID-19 vaccine in Korean children and adolescents aged 5 to 17 years
Seontae Kim, Yeseul Heo, Soon-Young Seo, Do Sang Lim, Enhi Cho, Yeon-Kyeng Lee
Osong Public Health and Research Perspectives.2022; 13(5): 382. CrossRef - COVID-19 Vaccination in Korea: Past, Present, and the Way Forward
Eliel Nham, Joon Young Song, Ji Yun Noh, Hee Jin Cheong, Woo Joo Kim
Journal of Korean Medical Science.2022;[Epub] CrossRef - Self-reported adverse events after 2 doses of COVID-19 vaccine in Korea
Yunhyung Kwon, Insob Hwang, Mijeong Ko, Hyungjun Kim, Seontae Kim, Soon-Young Seo, Enhi Cho, Yeon-Kyeng Lee
Epidemiology and Health.2022; 45: e2023006. CrossRef
- A Nationwide Survey of mRNA COVID-19 Vaccinee’s Experiences on Adverse Events and Its Associated Factors
- COVID-19 vaccine safety monitoring in the Republic of Korea: February 26, 2021 to April 30, 2021
- Hyun-kyung Oh, Eun Kyeong Kim, Insob Hwang, Tae Eun Kim, Yeon-kyeong Lee, Eunju Lee, Yeon-Kyeng Lee
- Osong Public Health Res Perspect. 2021;12(4):264-268. Published online August 13, 2021
- DOI: https://doi.org/10.24171/j.phrp.2021.0157
- 5,939 View
- 142 Download
- 7 Web of Science
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF - Objectives
On February 26, 2021, coronavirus disease 2019 (COVID-19) vaccination was started for high-priority groups based on the recommendation of the Advisory Committee on Immunization Practices with 2 available COVID-19 vaccines (AstraZeneca and Pfizer-BioNTech) in Korea. This report provides a summary of adverse events following COVID-19 vaccination as of April 30, 2021.
Methods
Adverse events following immunization are notifiable by medical doctors to the Korea Immunization Management System (KIMS) under the national surveillance system. We analyzed all adverse events reports following COVID-19 vaccination to the KIMS from February 26 to April 30, 2021.
Results
In total, 16,196 adverse events following 3,586,814 administered doses of COVID-19 vaccines were reported in approximately 2 months (February 26 to April 30, 2021). Of these, 15,658 (96.7%) were non-serious adverse events, and 538 (3.3%) were serious adverse events, including 73 (0.5%) deaths. The majority of adverse events (n=13,063, 80.7%) were observed in women, and the most frequently reported adverse events were myalgia (52.2%), fever (44.9%), and headache (34.9%). Of the 73 deaths following the COVID-19 vaccination, none were related to the vaccines.
Conclusion
By April 30, 3.6 million doses of the COVID 19 vaccine had been given in Korea, and the overwhelming majority of reports were for non-serious events. The Korea Disease Control and Prevention Agency continues to monitor the safety of COVID-19 vaccination. -
Citations
Citations to this article as recorded by- A prospective cohort study protocol: monitoring and surveillance of adverse events following heterologous booster doses of Oxford AstraZeneca COVID-19 vaccine in previous recipients of two doses of Sinopharm or Sputnik V vaccines in Iran
Shahin Soltani, Behzad Karami Matin, Mohammad Mehdi Gouya, Sayed Mohsen Zahraei, Ghobad Moradi, Omid Chehri, Moslem Soofi, Mehdi Moradinazar, Fatemeh Khosravi Shadmani, Mahsa Kalantari, Hamidreza Khajeha, Mohammad Hassan Emamian, Farid Najafi
BMC Public Health.2023;[Epub] CrossRef - Herpes Zoster Reactivation After mRNA and Adenovirus-Vectored Coronavirus Disease 2019 Vaccination: Analysis of National Health Insurance Database
Jin Gu Yoon, Young-Eun Kim, Min Joo Choi, Won Suk Choi, Yu Bin Seo, Jaehun Jung, Hak-Jun Hyun, Hye Seong, Eliel Nham, Ji Yun Noh, Joon Young Song, Woo Joo Kim, Dong Wook Kim, Hee Jin Cheong
The Journal of Infectious Diseases.2023; 228(10): 1326. CrossRef - Safety and effectiveness of BNT162b2 mRNA Covid-19 vaccine in adolescents
Young June Choe, Seonju Yi, Insob Hwang, Jia Kim, Young-Joon Park, Eunhee Cho, Myoungyoun Jo, Hyunju Lee, Eun Hwa Choi
Vaccine.2022; 40(5): 691. CrossRef - Direct and Indirect Associations of Media Use With COVID-19 Vaccine Hesitancy in South Korea: Cross-sectional Web-Based Survey
Minjung Lee, Myoungsoon You
Journal of Medical Internet Research.2022; 24(1): e32329. CrossRef - Self-Reported COVID-19 Vaccines’ Side Effects among Patients Treated with Biological Therapies in Saudi Arabia: A Multicenter Cross-Sectional Study
Lama T AlMutairi, Wesal Y Alalayet, Sondus I Ata, Khalidah A Alenzi, Yazed AlRuthia
Vaccines.2022; 10(6): 977. CrossRef - COVID-19 vaccine safety monitoring in Republic of Korea from February 26, 2021 to October 31, 2021
Insob Hwang, Kyeongeun Park, Tae Eun Kim, Yunhyung Kwon, Yeon-Kyeng Lee
Osong Public Health and Research Perspectives.2021; 12(6): 396. CrossRef
- A prospective cohort study protocol: monitoring and surveillance of adverse events following heterologous booster doses of Oxford AstraZeneca COVID-19 vaccine in previous recipients of two doses of Sinopharm or Sputnik V vaccines in Iran



 First
First Prev
Prev


